Malt intended for use in beer brewing or elsewhere in the food industry
The aliquot of an extract of malt is added to a buffered starch solution and allowed to stand for exactly 30 min at 20 °C. Then, the maltose – formed primarily from the starch through the action of the β-amylase – is measured using iodine and is determined according to the following chemical reaction:
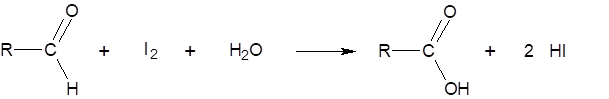
The method describes how to determine the free and total chlorine content using a titrimetric method with DPD.
The analysis involves a reaction with N,N-diethyl-1,4-phenylenediamine (DPD), which forms a compound possessing a red color at a pH of 6.2–6.5. The solution is titrated with an ammonium iron(II) sulfate standard solution until the red color disappears. Total chlorine is measured through the addition of potassium iodide, of which a known amount in excess of that required is added to the solution in advance.
Determination of the concentration of alkaline cleaners in sodium hydroxide (NaOH) and aluminum (Al).
All alkaline cleaning solutions or "stack solutions" that contain dissolved aluminum (Al3+) and/or soda (Na2CO3) in addition to sodium hydroxide (NaOH) as a cleaning component.
The sodium hydroxide bound as aluminate and titrated in the p-value determination is calculated by subsequent determination of the aluminum and deducted, as it is no longer available for purification.
The aluminum determination itself is based on the reaction between aluminum hydroxide and sodium fluoride, in which sodium hydroxide is released and thus serves as a measure for calculating the aluminum.
Carbonates interfering with the titration are precipitated with Ba2+ ions.
Determination of the concentration of alkaline cleaners in sodium hydroxide (NaOH), soda (Na2CO3) and aluminum (Al).
All alkaline cleaning solutions or "stack solutions" that contain dissolved aluminum (Al3+) and/or soda (Na2CO3) in addition to sodium hydroxide (NaOH) as a cleaning component.
The sodium hydroxide bound as aluminate and titrated in the p-value determination is calculated by subsequent determination of the aluminum and deducted, as it is no longer available for purification.
The aluminum determination itself is based on the reaction between aluminum hydroxide and sodium fluoride, in which sodium hydroxide is released and thus serves as a measure for calculating the aluminum.
Titration against methyl orange instead of the Tashiro indicator leads to incorrect results. When sodium fluorif (NaF) is added to the alkaline solution, hydrofluoric acid (HF) is formed. The hydrofluoric acid destroys the methyl orange indicator.
Consideration of phosphates and impurities, especially hydrolysis products of proteins in the concentration determination of alkaline cleaners on sodium hydroxide (NaOH) and soda (Na2CO3).
All alkaline cleaning solutions or "batch solutions" that contain soda (Na2CO3) as a cleaning component in addition to sodium hydroxide (NaOH).
Additives containing phosphoric acid in particular - but also other types of additive - can simulate a more or less significant Na2CO3 content due to their buffering effect. Firstly, phosphoric acid significantly blunts the p-value by forming sodium phosphate (Na3PO4) and secondly, the difference between the m-value and the p-value is increased because sodium dihydrogen phosphate (NaH2PO4) is largely formed from disodium hydrogen phosphate (Na2HPO4) in the pH range 8.2 to 4.3. The same applies - but to a lesser extent due to the molecular sizes - to typical phosphonic acids or their salts, but not, for example, to most surfactants.
To determine the exact concentration or pH value, the content of buffering substances - phosphates and impurities - must be taken into account in addition to the degree of carbonation, as otherwise the concentration of the active cleaning agent will be too high.
Determination of the p-value of the detergent solution with an acid solution (HCl or H2SO4) with corresponding normality up to the color change of phenolphthalein (pH 8.2).
NaOH + HCl → NaCl + H2O colorless against phenolphthalein
2 NaOH + H2SO4 → Na2SO4 + 2 H2O
Determination of the soda ash (Na2CO3) content of the detergent solution with an acid solution (HCl or H2SO4) with corresponding normality up to the color change from methyl orange (pH 4.3).
Na2CO3 + HCl → NaHCO3 + NaCl
NaHCO3 + HCl → NaCl + H2O + CO2 yellowish brown against methyl orange
2 Na2CO3 + H2SO4 → 2 NaHCO3 + Na2SO4
2 NaHCO3 + H2SO4 → 2 Na2SO4 + H2O + 2 CO2
The most important step is the expulsion of CO2. This is to ensure that no more hydrogen carbonates or carbonates can form during the subsequent back titration with NaOH. For this purpose, a significant acidification below pH 4.3 must be carried out with acid in order to be able to drive out CO2 using an inert gas.
The proportion of buffering substances can then be determined by back titration with NaOH to pH 4.3 or 8.2.
Determination of the concentration of dissolved carbon dioxide in carbonated beverages through titrimetry (dimensional analysis)
This method is suitable for determining the dissolved carbon dioxide in carbonated beverages for concentrations ranging from 0 to 8.4 g/l.
Through the addition of a sodium hydroxide solution, the carbon dioxide in beer becomes bound as sodium hydrogen carbonate or sodium carbonate. Sulfuric acid is added to an aliquot of the beer treated with sodium hydroxide. This causes the carbon dioxide to be released again, after which a stream of air conducts the carbon dioxide into a barium hydroxide solution. Through titration of the excess barium hydroxide, the carbon dioxide content of the beer can be determined [1].